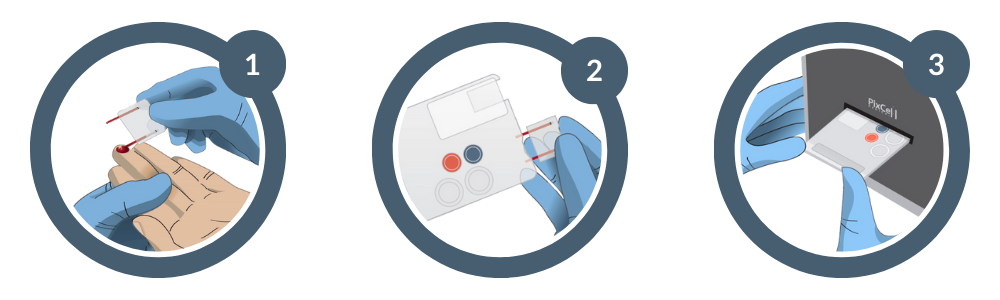
Request More Info
Want to learn more about PixCell? No problem - fill in your details below and our team will be in touch!
Thank you
for getting in touch!
We appreciate you contacting us. One of our
colleagues will get back in touch with you soon!Have
a great day!